Drug testing kits: where false positives usually come from
Author
Date Published
Reading Time

False positives in drug testing kits often stem from cross-reactive medications, poor sample handling, expired reagents, and operator error rather than actual substance use. For safety managers, buyers, and frontline users comparing drug testing kits with wholesale alcohol breathalyzers or other workplace screening tools, understanding these common causes is essential to improve accuracy, protect compliance, and make better procurement decisions.
Why do drug testing kits return false positives in real workplace screening?
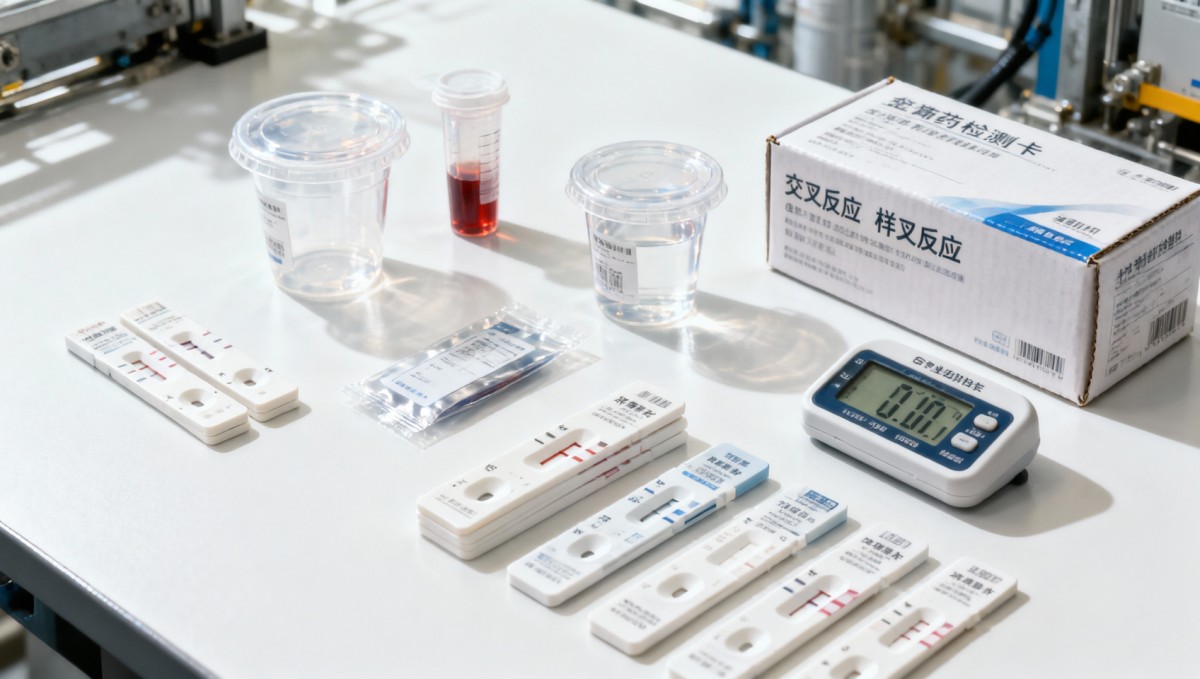
In industrial settings, a false positive is rarely a single-point failure. It usually comes from a chain of small weaknesses across the sample, the kit, the operator, and the environment. For employers running routine screening every week, every month, or before site access, one incorrect result can delay onboarding, interrupt shift planning, and create avoidable compliance disputes.
Most rapid drug testing kits used in field programs rely on immunoassay screening. These kits are designed for speed, not final legal confirmation. That distinction matters. A screening result indicates that the assay detected a target or a cross-reactive compound above a preset cutoff threshold. It does not automatically prove substance misuse, especially when medications, supplements, or sample interference are present.
For procurement teams, this is where product evaluation often goes wrong. Buyers may compare only unit price, panel count, or delivery lead time of 7–15 days, while missing the operational variables that shape accuracy. In safety-sensitive sectors such as construction, utilities, logistics, and plant maintenance, reliability depends on the full testing workflow rather than the strip alone.
Global Industrial Core supports this broader view by linking workplace screening decisions to safety operations, measurement discipline, and compliance logic. For decision-makers, the practical question is not whether false positives can happen. They can. The real question is which causes are predictable, controllable, and preventable before they disrupt labor management or trigger formal review.
The 4 most common source categories
- Cross-reactivity: certain prescription drugs, over-the-counter medicines, and supplements may react with assay antibodies and mimic a target substance.
- Sample handling problems: mislabeled cups, delayed reading windows, contamination, or storage outside the specified temperature range can distort results.
- Reagent or kit condition: expired kits, moisture exposure, damaged packaging, and unstable warehouse conditions can degrade performance.
- Operator error: incorrect timing, poor interpretation of faint lines, and inadequate chain-of-custody practices remain frequent issues in decentralized testing.
These categories matter because each one requires a different control measure. Some are solved through better specifications, some through training, and some through a documented confirmation process. A robust screening program usually works in 3 layers: validated kit selection, controlled administration, and laboratory confirmation for non-negative results.
Which factors create the highest risk of false positives during daily use?
In day-to-day operations, medication cross-reactivity is often the first suspect. Pain relievers, cold remedies, sleep aids, and some prescription medications may interfere with rapid drug testing kits depending on the panel design and assay chemistry. This is especially relevant in industrial workforces where employees may use legitimate medications for fatigue, injury recovery, seasonal illness, or chronic conditions.
The second major factor is timing discipline. Many rapid kits require interpretation within a defined reading window, commonly a few minutes after application. Reading too early can produce incomplete development. Reading too late may create evaporation effects or ambiguous line intensity. For operators managing multiple samples in one session, even a 2–5 minute timing error can change the practical outcome.
Storage conditions are another neglected variable. Drug testing kits are usually expected to remain within the manufacturer’s stated temperature and humidity conditions before use. In field procurement, products may pass through a distributor, a warehouse, and a remote site office before opening. If kits spend days in overheated vehicles or damp storage rooms, performance consistency may drop even before the expiry date.
Sample integrity also matters. Urine samples, oral fluid samples, and other matrices each have handling sensitivities. A mislabeled specimen, a poorly sealed cup, a contaminated collection area, or a chain-of-custody gap can create uncertainty. When a business operates across 3 shifts or multiple project sites, small procedural inconsistencies scale quickly into procurement complaints and HR escalation.
Risk matrix for common false positive sources
The table below helps buyers and program managers compare where false positive risk most often emerges and what type of control reduces it most effectively.
This comparison shows that many false positives are process-related rather than purely product-related. That distinction is critical for buyers evaluating supplier performance. A low complaint rate often depends on matching the kit to the operating environment and the capability of the staff who administer it.
Why alcohol breathalyzers and drug testing kits should not be judged the same way
Some organizations compare drug testing kits directly with wholesale alcohol breathalyzers because both serve workplace screening. However, the interference profile differs. Breath alcohol screening is generally narrower in target scope, while multi-panel drug testing kits may screen 5, 10, or 12 analyte groups in one device. The more complex the panel, the greater the need for disciplined interpretation and follow-up confirmation.
That does not make rapid drug screening unsuitable. It means the procurement model should account for panel design, matrix type, training burden, and confirmation pathway. For enterprise buyers, this is a systems decision, not a disposable-goods decision.
How should buyers compare drug testing kits before procurement?
A sound procurement review starts with intended use. Pre-employment screening, random workplace testing, post-incident checks, and return-to-duty programs do not always require the same panel configuration or workflow. Buyers should first define 3 core variables: sample type, panel scope, and decision urgency. This avoids overbuying complex kits for simple screening or underbuying for regulated environments.
The second step is to look beyond headline sensitivity claims. Ask how the kit performs under ordinary site conditions, how easy it is to interpret faint lines, and what storage control is required. A product with acceptable laboratory performance but weak field usability may increase operational disputes. For many procurement teams, reduced retest burden is more valuable than a slightly lower carton price.
Documentation also deserves attention. Buyers should request instructions for use, batch traceability details, storage guidance, and any available conformity or quality system references relevant to the supply chain. In B2B screening programs, documentation quality often predicts service reliability. If a supplier cannot explain handling limits, cutoff logic, or shelf-life management clearly, downstream confusion is likely.
Global Industrial Core approaches sourcing questions from an industrial operations perspective. That means evaluating not only unit economics, but also consistency, compliance workflow, and deployment friction across multisite environments. For EPC contractors, facility managers, and procurement directors, the right kit is the one that fits the real program design and can be supported over 2–4 quarters without process drift.
Practical procurement checklist
- Confirm whether the program requires urine, oral fluid, or another sample type based on site practicality and policy.
- Match the panel count to real risk exposure rather than choosing the widest panel by default.
- Review shelf life, storage range, and packaging resilience for transport across warehouse and field conditions.
- Ask about operator training needs, reading window control, and support for non-negative confirmation workflows.
- Check whether the supplier can support sample orders, batch documentation, and replenishment planning.
Buyer comparison table for selection decisions
The next table can be used during vendor review meetings to compare drug testing kits on criteria that matter to operators, purchasing teams, and compliance managers.
Used correctly, this table helps separate low-price offers from low-risk offers. For many enterprises, the total cost of a disputed result, retest, delayed site access, or HR case review is far higher than the saving gained from choosing the cheapest screening kit.
What controls reduce false positives after the kits arrive on site?
Once drug testing kits are delivered, the focus shifts from product choice to implementation discipline. A practical site program should define who stores the kits, who checks batch dates, who conducts the test, and who reviews non-negative outcomes. When these roles are vague, errors multiply. Even a well-selected kit can generate disputes if operational ownership is unclear across departments.
The first control is environmental management. Kits should be kept in accordance with the supplier’s stated storage conditions, away from direct heat, moisture, and uncontrolled transport exposure. Inventory should be rotated by batch, with routine checks at least monthly. If a site has harsh ambient conditions, buyers should confirm whether secondary packaging or controlled storage cabinets are needed.
The second control is procedure standardization. Frontline users need clear instructions on specimen collection, labeling, timing, reading windows, and escalation steps. A 4-step workflow is often effective: collect, verify, read within the specified time, and document. Standardized forms reduce ambiguity, especially when multiple supervisors or contractors share responsibility on one project.
The third control is confirmation management. Screening kits are useful for rapid decisions, but non-negative findings should move into a defined review path. In many workplaces, that means immediate documentation, medication disclosure review, and laboratory confirmation where policy requires it. This reduces the risk of disciplinary action based only on a presumptive screening result.
Implementation steps that reduce disputes
- Create a written testing SOP covering storage, use conditions, timing, and result escalation.
- Train operators before launch and refresh skills every 3–6 months when turnover is high.
- Use batch logs and expiry reviews to prevent use of outdated or compromised inventory.
- Separate screening from final adjudication and define when laboratory confirmation is mandatory.
Compliance and documentation points decision-makers often overlook
For corporate decision-makers, the testing kit itself is only one part of defensible screening. Policies should align with local labor rules, internal safety standards, and any contractual site requirements. If an employer operates internationally, even simple issues like consent forms, result handling, and retention periods can vary. Procurement therefore needs coordination with HR, legal, safety, and site operations.
This is where a technical sourcing partner adds value. Global Industrial Core helps industrial buyers connect product comparison with operational controls, certification awareness, and deployment practicality. That is especially useful when screening tools are procured alongside safety devices, measurement instruments, or alcohol breathalyzers under one broader workplace risk-management budget.
FAQ: what do users, buyers, and managers ask most often?
The questions below reflect common search intent from information researchers, operators, procurement staff, and business leaders who need practical answers before selecting or using drug testing kits in workplace programs.
Can over-the-counter medicine cause a false positive on drug testing kits?
Yes, it can happen. Some over-the-counter products and prescription medicines may cross-react with immunoassay screening. That is why a non-negative rapid test should not be treated as final proof on its own. The safer practice is to collect medication disclosure information and follow a confirmation process when policy or risk level requires it.
How often should stored kits be checked?
A monthly review is a practical minimum for most sites, especially when stock is held in more than one location. High-turnover or remote operations may need more frequent checks. Review expiry dates, packaging condition, and whether the storage environment stayed within the stated range during the previous 30 days.
Are more panel options always better?
Not necessarily. A 10-panel or 12-panel configuration may be useful in some programs, but unnecessary breadth can increase complexity, review burden, and questions around cross-reactivity. Buyers should choose the narrowest panel that still matches actual workplace risk, policy requirements, and the intended use case.
What should procurement teams ask suppliers before ordering?
Ask about intended use, sample type, storage conditions, shelf life, batch traceability, reading window, documentation package, sample availability, and support for dispute handling. Also ask about standard lead time, which in many supply chains may range from 1–3 weeks depending on order volume and destination. These questions reduce surprises after delivery.
Why work with Global Industrial Core when evaluating workplace screening tools?
Workplace screening is not just a purchasing task. It sits at the intersection of safety, measurement discipline, compliance risk, and operational continuity. Global Industrial Core helps industrial buyers assess drug testing kits in the same structured way they evaluate other mission-critical tools: by looking at field conditions, documentation quality, implementation burden, and supply reliability together rather than in isolation.
For information researchers, we help narrow the shortlist. For operators, we highlight usability and error prevention. For procurement teams, we clarify comparison criteria and sourcing risks. For enterprise decision-makers, we connect the purchase to broader site governance, contractor management, and workforce screening policy. This cross-functional view is especially useful when comparing drug testing kits with wholesale alcohol breathalyzers or other safety-control equipment.
If you are planning a new screening program or reviewing an existing one, you can contact Global Industrial Core for practical support on panel selection, storage considerations, expected lead time, documentation review, sample support, and supplier comparison. We can also help map your requirements across 3 key decision areas: product fit, operational workflow, and compliance handling.
Contact us when you need a clearer basis for quotation requests, parameter confirmation, replacement options, batch planning, or a more defensible procurement specification. That is the fastest way to reduce avoidable false positives, improve screening consistency, and make better purchasing decisions before the next order cycle begins.
Technical Specifications
Expert Insights

Chief Security Architect
Dr. Thorne specializes in the intersection of structural engineering and digital resilience. He has advised three G7 governments on industrial infrastructure security.
Related Analysis
- Apr 14, 2026How accurate is a true RMS multimeter on VFD signals?Author :Precision Metrology Expert
- Apr 14, 2026When a digital oscilloscope wholesale quote looks cheap at firstAuthor :Precision Metrology Expert
- Apr 14, 2026What changes when an ozone generator goes commercial gradeAuthor :Environmental Engineering Director
Core Sector // 01
Security & Safety







